Collaboration Brings Rapid At-Home STI Testing with Virtual Follow-Up
Posted on 20 Apr 2026
Sexually transmitted infections (STIs) are often asymptomatic, and reliance on clinic-based testing can delay diagnosis and treatment. More than one million curable infections are acquired daily worldwide among people aged 15–49, and complications contribute to infertility in nearly 100,000 women in the United States each year. Faster access to accurate diagnostics remains a critical clinical need. Addressing this need, a new partnership will expand access to an at-home PCR test that delivers lab-quality results in about 30 minutes with integrated virtual follow-up.
Visby Medical’s Women’s Sexual Health PCR test, an U.S. Food and Drug Administration (FDA)-authorized at-home PCR STI test, will be offered through a collaboration with Wisp, a U.S. women’s telehealth platform. The program will begin with targeted patient outreach, with plans to expand access over time. When results are positive for chlamydia, gonorrhea, or trichomoniasis, patients are referred to Wisp for follow-up telehealth care.
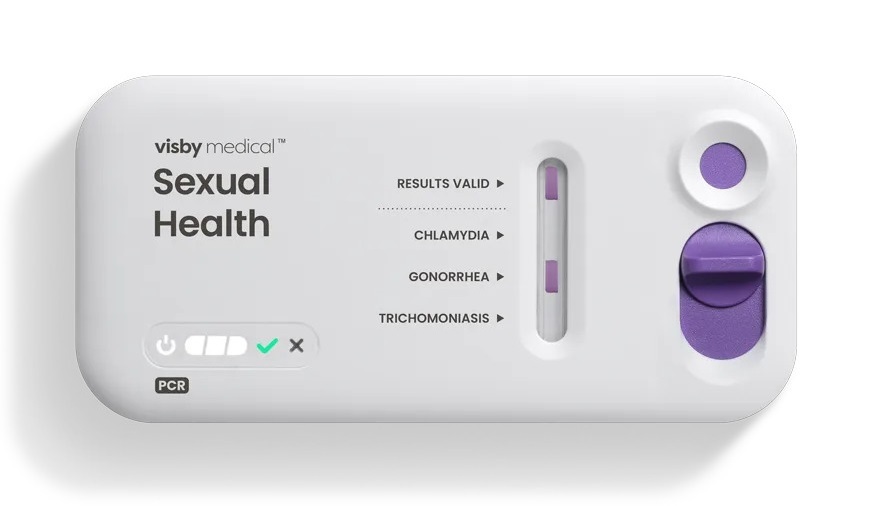
The palm-sized device performs PCR and reports results within 30 minutes via Visby’s mobile app. The integrated model is intended to streamline the path from diagnosis to treatment compared with conventional care pathways. For those who test positive, Wisp provides a complimentary virtual consultation with a licensed provider, and antibiotics can be prescribed from home when clinically appropriate.
The collaboration emphasizes discreet access to testing and timely clinical guidance. Wisp’s platform supports secure online consultations, personalized care plans, and confidential treatment pathways. The at-home test is also eligible for flexible spending account and health savings account reimbursement, and the partners plan to broaden availability while continuing to reduce time between diagnosis and treatment.
“This partnership came directly from listening to our patients,” said Monica Cepak, CEO of Wisp. “Women are asking for faster answers and more seamless next steps. By pairing Visby’s FDA-authorized at-home PCR test with Wisp’s care model, we’re delivering on our mission to support women across every life stage with fast, private, and stigma-free care.”
“By providing timely medical advice through telehealth, the value of the Visby at-home sexual health test significantly increases,” said Dr. Gary Schoolnik, Chief Medical Officer at Visby Medical and Professor of Medicine at Stanford Medical School. “This ensures that women who use the Visby test will receive prompt and effective advice and a path to treatment, if indicated.”