Novel Nanosensing Technologies Developed for Exosome Detection and Profiling
By LabMedica International staff writers
Posted on 20 Nov 2019
Exosomes are extracellular vesicles (EVs) that are produced in the endosomal compartment of most eukaryotic cells. Liquid biopsy technologies have been developed to do high-throughput exosome protein profiling and point-of-care (POC) exosome analysis.Posted on 20 Nov 2019
The aim is to develop multiplexed assay systems to streamline the analysis of exosomes and evaluate their clinical utility for cancer management, both as diagnostic tools and to predict disease recurrence. Nanoplasmonic sensors are fabricated to accommodate at most one exosome and individually imaged in real time, enabling the label-free recording of digital responses in a highly multiplexed geometry.
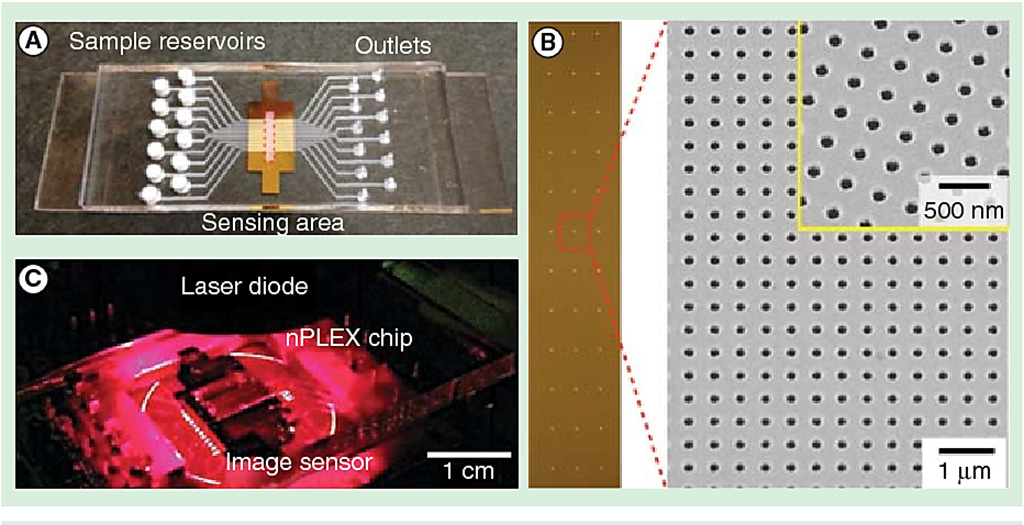
Image: The nano-plasmonic exosome (nPLEX) sensor (Photo courtesy of Massachusetts General Hospital).
Scientists at the Massachusetts General Hospital (MGH, Boston, MA, USA) and their colleagues developed the nano-plasmonic exosome (nPLEX) sensor, in routine use at MGH, which detects exosome protein levels based on “spectral shifts” (intensity changes) of light through thousands of optimally spaced 200-nanometer (nm) holes. Antibodies against cancer biomarkers get immobilized on the nanopores for capture.
Nanoholes on the sensor are gold and exosomes are labeled by gold nanoparticles for signal amplification. Gold has proven to have the best chemical stability for this type of assay. The device is being manufactured on a glass-based substrate, as silicon wafers proved to be too fragile. The iMEX (integrated magnetic-electrochemical exosome) device, meanwhile, integrates vesicle isolation and detection in a single platform. Target-specific exosomes first get enriched through immunomagnetic selection, for high detection sensitivity. The sensors can be miniaturized and expanded for parallel measurements.
The nPLEX has demonstrated good accuracy and 100 times greater sensitivity than the commonly used enzyme-linked immunosorbent assay (ELISA) and able to detect as few as 3,000 exosomes. The team has also come up with a protein signature for pancreatic ductal adenocarcinoma (PDAC) that is powerful enough for diagnostic purposes, a combination of EGFR (epidermal growth factor receptor), EpCAM, MUC1, GPC1 and WNT2. As demonstrated in a prospective study involving more than 100 patients and age-matched controls, as the tumors shrink the biomarker combination gets lower and lower.
The scientists have also used nPLEX technology to show how exosome protein expression of cancer cells changes with drug treatment. Discovery of these unique, drug-dependent protein signatures suggests the exosome screening assay will be a potentially powerful molecular screening tool. Hyungsoon Im, PhD, an assistant professor of radiology and a senior author of the study, said, “The screening assay could be incorporated into point–of–care (POC) instruments for more rigorous testing of different drugs in different settings. The study was presented at the 2019 Next Generation Dx Summit held August 20-22, 2019 in Washington, DC, USA.
Related Links:
Massachusetts General Hospital














