Breath Analysis Identifies Cancer Patients Likely to Benefit from Immunotherapy
By LabMedica International staff writers
Posted on 23 Sep 2019
An “electronic nose” analytical device has been developed that can determine the likelihood of a cancer patient to respond successfully to immunotherapy.Posted on 23 Sep 2019
Immune checkpoint inhibitors such as nivolumab and pembrolizumab have improved the survival outcome of advanced non-small-cell lung cancer (NSCLC) patients. However, most patients do not benefit from this treatment. Therefore, biomarkers are needed that can accurately predict the patient’s response.
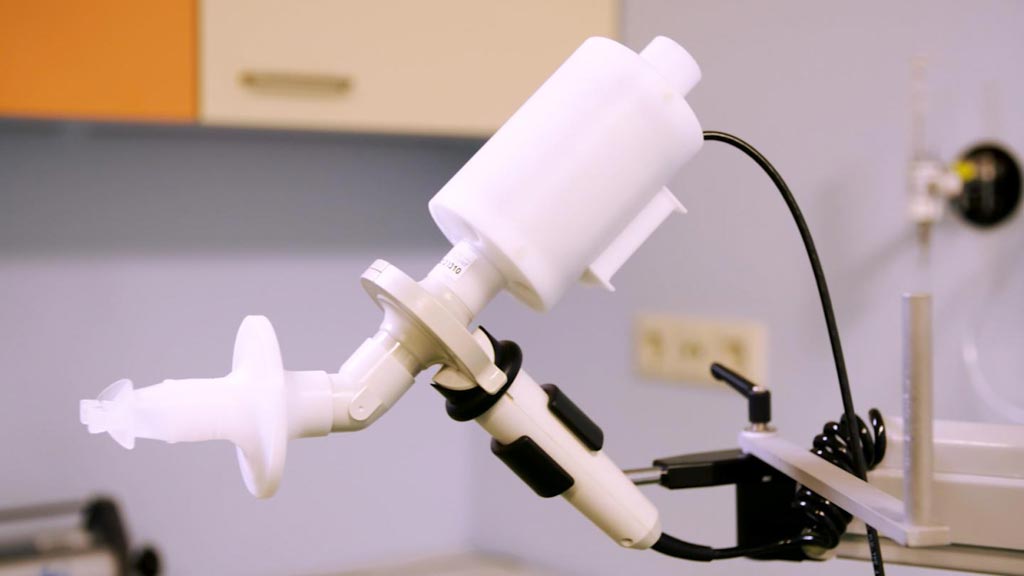
Image: Breath analysis of lung cancer patients with the eNose device could identify patients who would or would not respond to immunotherapeutic treatment with 85% accuracy (Photo courtesy of Amsterdam University Medical Centers).
Investigators at the Radboud University Medical Centre (Nijmegen, The Netherlands) and their collaborators at other institutions hypothesized that molecular profiling of exhaled air may capture the inflammatory milieu related to the individual responsiveness to anti-programmed death ligand 1 (PD-1) therapy. The current study aimed to determine the accuracy of exhaled breath analysis for assessing nonresponders versus responders to anti-PD-1 therapy in NSCLC patients.
The electronic nose (eNose) device comprised a metal oxide semiconductor electronic sensor positioned at the rear end of a pneumotachograph. A pneumotachograph is a type of respirometer that is used to assess pulmonary function. This instrument can measure the mechanical function of lungs, chest wall, and respiratory muscles by recording volume, flow, and pressure changes during expiratory or inspiratory events. The eNose sytem used for this study was produced by the biotechnology company Breathomix (Reeuwijk, The Netherlands).
The purpose of the eNose was to detect volatile organic compounds (VOCs), which are present in about 1% of exhaled breath. The investigators speculated that the mix of VOCs in the breath of patients with advanced NSCLC might indicate whether or not the patient would respond to anti-PD1 therapy. The measurement took less than a minute, and the results were compared to an online database where machine-learning algorithms immediately identified whether or not the patient was likely to respond to anti-PD1 therapy.
For the study, the investigators worked with 143 patients with advanced NSCLC. The eNose device was used to establish the breath profiles of the patients two weeks before they began treatment with nivolumab or pembrolizumab. After three months the investigators used standard criteria (Response Evaluation Criteria of Solid Tumors, RECIST) to assess whether the patients were responding to the treatment nor not. Results from the first 92 patients (who started treatment between March 2016 and February 2017) were validated by the results from the remaining 51 patients (who started treatment after April 2017).
Results revealed that eNose analysis of the breath of lung cancer patients could identify with 85% accuracy those who would or would not respond to immunotherapeutic treatment. This finding could potentially prevent the application of ineffective treatment for patients identified as probable nonresponders.
Senior author Dr. Michel van den Heuvel, professor of thoracic oncology at the Radboud University Medical Centre, said, "The introduction of immunotherapy has dramatically improved the treatment of advanced stage non-small cell lung cancer but unfortunately it is only effective in a subset of patients, which was about 20% when we started the study. Currently, there is no test available that can accurately predict who will benefit from this treatment, apart from PD-L1 testing by immuno-histochemistry. This is today's biomarker of choice, despite its analytic and predictive limitations, when making clinical decisions about whether or not to treat a patient with immunotherapy."
"We are convinced that this study merely scratches the surface," said Dr. van den Heuvel. "It represents the first introduction of modern precision medicine, namely that molecular fingerprints can be easily obtained and quickly analyzed on the spot. This truly offers new possibilities for the individual patient and the doctor. The power of this eNose system is that it has been properly validated, both technically and clinically, which is essential. We believe that analysis of exhaled breath is going to become an important diagnostic tool and will guide future treatment in oncology as well as in many other diseases."
The study was published in the September 17, 2019, online edition of the journal Annals of Oncology.
Related Links:
Radboud University Medical Centre
Breathomix














