Novel Chemiluminescent Immunoassay Detects Toxoplasma in Human Sera
By LabMedica International staff writers
Posted on 26 Jul 2016
Toxoplasmosis is common disease in humans and animals caused by the protozoan parasite Toxoplasma gondii and while this disease in healthy people is usually asymptomatic, in congenital cases and immune deficient patients it can lead to serious pathological effects.Posted on 26 Jul 2016
Currently, routine diagnosis of toxoplasmosis relies mainly on the use of various serological tests to detect specific antibodies in the serum samples of infected patients. The outcome of these tests depends on the antigen types and the methods of detection, and often the early recognition of the infection or precise distinction between phases of toxoplasmosis is difficult.

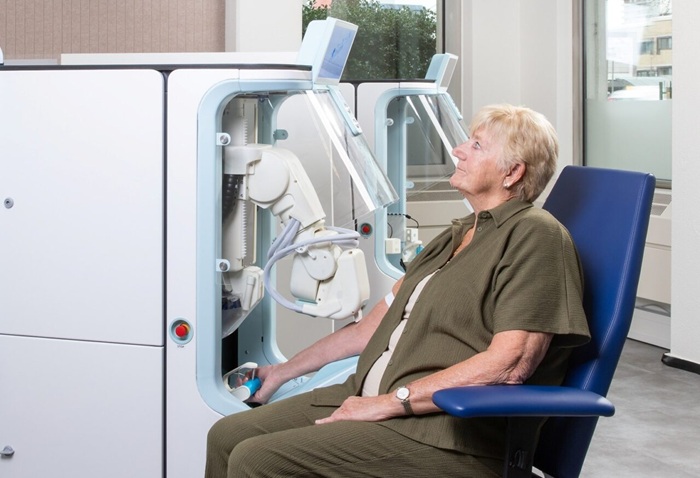
Image: The Centro XS³ LB 960 microplate luminometer (Photo courtesy of Berthold Technologies).
Scientists at the Gdansk University of Technology (Poland) developed new immunochemical reagents in the form of original chemiluminescence (CL) labels, which enabled them to perform the immunoglobulin G (IgG) chemiluminescent immunoassay (CLIA) tests for diagnosis of human toxoplasmosis. To assess the performance of novel immunochemical reagents in the CLIA assay, SAG2-GRA1-ROP1L chimeric protein for the detection of IgG specific antibodies was utilized. The latter was previously tested as diagnostic antigen in typical enzyme-linked immunosorbent assay (ELISA). The emission profiles were simultaneously measured over 3 to 30 second periods to collect at least 95% of the CL signal, using a Centro XS3 LB-960 microplate luminometer (Berthold Technologies, Bad Wildbad, Germany).
All of the sera used in the study were received from a routine toxoplasmosis screening. A total of 92 sera were analyzed, using commercial tests (Vidas Toxo-IgG II, Toxo-Screen DA, and Vidas Toxo-IgM; bioMérieux, Marcy l'Etoile. France) and divided into the two groups: group I (IgG positive and IgM positive or negative) included 47 sera from patients with toxoplasmosis and group II (IgG and IgM negative) consisted of 45 serum samples from seronegative individuals. The diagnostic usefulness of variously acridinium ester (AE)-labeled antibodies was tested on the same day. The scientists found that the assay with the participation of AE1 conjugate expressed greater ability to distinguish the sera from patients with T. gondii infection and healthy individuals.
The authors concluded that with the development and application of recombinant antigen (rAg) and recombinant antibody (rAb) technology as well as the use of a novel acridinium CL labels, CLIA approach can acquire a new dimension. However, further work is needed before this kind of immunoassay will be available for clinical purposes. The study was published online on May 24, 2016, in the journal Diagnostic Microbiology and Infectious Diseases.
Related Links:
Gdansk University of Technology
Berthold Technologies
bioMérieux