New Test Accurately Detects Latent HIV
By LabMedica International staff writers
Posted on 14 Jun 2017
The ability of the human immunodeficiency virus (HIV) to lie dormant in a "reservoir" of CD4 cells has been the main obstacle to finding a cure; once a patient starts antiretroviral therapy, it becomes very important to measure the level of viremia, and whether the virus can replicate.Posted on 14 Jun 2017
Most tests available for detecting the virus are not very cost effective and take a lot of time. The most widely available test at the moment is the "quantitative viral outgrowth assay" (Q-VOA), but it requires large amounts of blood, is very labor intensive, and is quite expensive and additionally, the Q-VOA may also underestimate the amount of virus left.
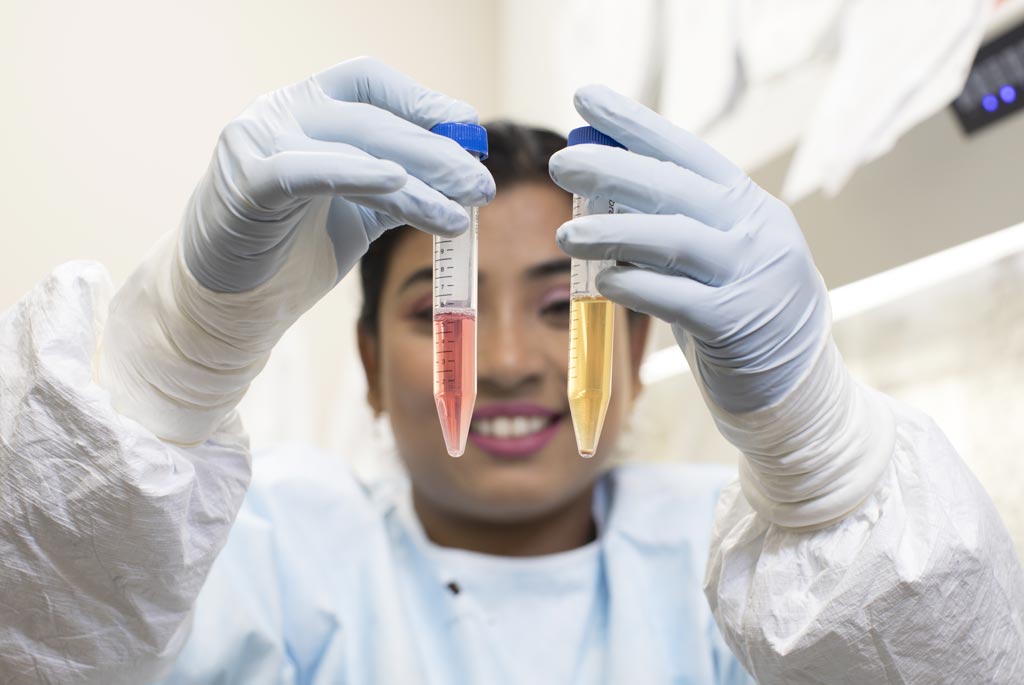
Image: Anwesha Sanyal, PhD, indicates HIV-infected cells collecting at the bottom of a test tube being prepared for the TZA test. The yellow color indicates the stimulated (Photo courtesy of the University of Pittsburgh).
Scientists at the University of Pittsburgh (PA, USA) and their colleagues developed a sensitive assay that can accurately and rapidly quantify inducible, replication-competent latent HIV-1 from resting CD4+ T cells, which is essential for HIV-1 eradication studies. The developed test is called TZA and it works by detecting a gene that is turned on only when replicating HIV is present, thereby flagging the virus for technicians to quantify.
The TZA test produces results in one week compared to the two weeks needed using the Q-VOA, and at a third of the cost. The TZA test also may be useful for quantification of replication-competent HIV-1 in the pediatric population, because of its low cell requirement, as well as in the lymph nodes and tissues where the virus persists. The assay has several advantages over existing technology in that it is sensitive; requires only a small blood volume; is faster, less labor intensive, and less expensive; and can be readily adapted into a high-throughput format. Using this assay, the authors show that the size of the inducible latent HIV-1 reservoir in aviremic participants on therapy is approximately 70-fold larger than previous estimates.
Phalguni Gupta, PhD, a professor and senior author of the study, said, “Using this test, we demonstrated that asymptomatic patients on antiretroviral therapy carry a much larger HIV reservoir than previous estimates, as much as 70 times what the Q-VOA test was detecting. Because these tests have different ways to measure HIV that is capable of replicating, it is likely beneficial to have both available as scientists strive toward a cure." The study was published on May 29, 2017, in the journal Nature Medicine.
Related Links:
University of Pittsburgh














