Test Confirms Presence of Human T-Cell Lymphotropic Virus-I/II Antibodies
By LabMedica International staff writers
Posted on 30 Dec 2014
A supplemental test for Human T-cell Lymphotropic Virus-I/II (HTLV-I/II) has been licensed and is intended for use as an additional, more specific test for human serum or plasma specimens that have previously tested positive on other HTLV-I/II blood donor screening tests.Posted on 30 Dec 2014
The Human T-cell Lymphotropic viruses (HTLV) are a group of human retroviruses known to cause diseases such as adult T-cell leukemia/lymphoma which is a rare form of blood cancer and inflammation of the nerves in the spinal cord, known as myelopathy, as well as other conditions such as uveitis, and infectious dermatitis.
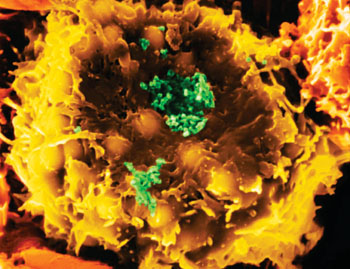
Image: Scanning electron micrograph of Human T-cell Lymphotropic Virus-1 (HTLV-I) (green) infecting a human T-lymphocyte (yellow) (Photo courtesy of Dr. Dennis Kunkel).
HTLV can be transmitted from person to person through breastfeeding, unprotected sexual contact, or transfusion of blood from an infected donor. The US Food and Drug Administration (FDA; Silver Springs, MD, USA) which has approved the test require that donated blood be tested for HTLV-I/II antibodies. Currently there are two FDA-licensed screening tests for HTLV-I/II. If the test is positive, the donation is discarded and the donor is notified of his or her deferral.
The MP Diagnostics HTLV Blot 2.4 (MP Biomedicals Asia Pacific Pte Ltd.; Singapore), is a qualitative enzyme immunoassay test, that provides blood establishments with additional information to convey to the donor; specifically, the test can confirm HTLV infection and determine which virus type is causing the infection, HTLV-I or HTLV-II. Many people who are infected with HTLV are unaware of the infection because the virus may not cause any symptoms or signs of infection. Additionally, many people infected with HTLV-I or HTLV-II may never develop any disease caused by the viruses. However, these asymptomatic carriers can still transmit the viruses to others.
Karen Midthun, MD, director of FDA's Center for Biologics Evaluation and Research, said, “The approval of MP Diagnostics HTLV Blot 2.4 will help blood establishments better counsel donors who have had positive results on an FDA-licensed HTLV-I/II screening test.”
Related Links:
US Food and Drug Administration
MP Biomedicals Asia Pacific Pte Ltd.














