Cerebral Malaria Biomarker Quantified
By LabMedica International staff writers
Posted on 17 Jul 2014
The detection, semi-quantification, and clinical use of the Plasmodium falciparum histidine-rich protein-2 (PfHRP-2) as a parasite antigen biomarker for cerebral malaria had been investigated.Posted on 17 Jul 2014
The examination of the cerebrospinal fluid (CSF) in cerebral malaria cases typically shows normal parameters with normal protein, glucose, and low cell count and hitherto a CSF biomarker for cerebral malaria has yet to be identified.
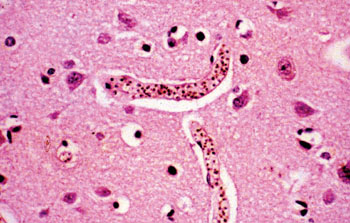
Image: Histology of a brain from a patient with cerebral malaria showing sequestration of parasites, pigmentation and obstruction of blood vessels (Photo courtesy of Chulalongkorn University).
An international team of scientists led by those from the Johns Hopkins Bloomberg School of Public Health (Baltimore, MD, USA) collected 73 CSF samples from Tanzanian children, 6 months to 9 years of age with P. falciparum parasitaemia. The CSF samples from nine Tanzanian children, who were microscopy negative for P. falciparum in peripheral blood, were also collected at the same site from 1994 to 1995. An additional 24 CSF samples were selected from archived CSF samples obtained from patients with neuroimmunological or neuroinflammatory conditions to use as controls.
The rapid diagnostic test (RDT) Binax NOW Malaria Test (Alere; Waltham, MA, USA), which detects both PfHRP-2 and panspecies aldolase was performed on the CSF samples. An immuno-polymerase chain reaction (PCR) was optimized and the real-time PCR amplifications were performed in a C1000 Thermal Cycler (Bio-Rad; Hercules, CA, USA). Western blots analysis was performed to compare some plasma and CSF samples.
The PfHRP-2 was detected in archival CSF samples from cerebral malaria patients from Tanzania both by a newly developed sensitive and specific immuno-PCR in 72 of 73 samples, and by rapid diagnostic tests in 62 of 73 samples. The geometric mean PfHRP-2 CSF concentration was 8.76 ng/mL with no significant differences in those who survived with 9.2 ng/mL, those who died with11.1 ng/mL, and those with neurologic sequelae with 10.8 ng/mL. The cerebral biomarker CSF PfHRP-2 was undetectable in all aparasitemic endemic and non-endemic control samples.
The authors concluded that the widely used RDTs for malaria may be useful in CSF detection of PfHRP-2 in patients with cerebral malaria. A total of 62 of 73 samples had at least 1 ng/mL PfHRP-2 concentration detectable, an amount well above the 100 pg limit of detection threshold, if 100 µL is used for the RDT. The CSF PfHRP2 is proportional to plasma such that detection in the CSF is reflective of elevated plasma or whole blood PfHRP2 seen in many studies of cerebral malaria. The study was published June 30, 2014, in the American Journal of Tropical Medicine and Hygiene.
Related Links:
Johns Hopkins Bloomberg School of Public Health
Alere
Bio-Rad














