Antinuclear-Antibody Testing Analyzed by Automated Indirect Immunofluorescence
By LabMedica International staff writers
Posted on 15 Apr 2014
Automated quantitative reading of fluorescence intensity has been evaluated for clinical relevance which allows for value-added reporting of test results. Posted on 15 Apr 2014
Antinuclear antibodies (ANA) are important diagnostic markers for systemic rheumatic diseases (SRD), such as systemic lupus erythematosus (SLE), systemic sclerosis (SSc), primary Sjögren’s syndrome (SS), mixed connective tissue disease (MCTD), and, to a lesser extent, polymyositis (PM), and dermatomyositis (DM).
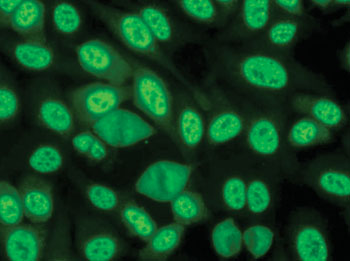
Image: Fluorescence antinuclear antibody speckled pattern staining (Photo courtesy of the Autoantibody Standardization Committee).
Medical laboratory scientists at the University Hospitals Leuven (Belgium) collected blood samples from 434 controls which included 150 healthy blood donors, 150 chronic fatigue syndrome, and 134 diseased controls, and 252 samples obtained at diagnosis from patients with SRD. This latter group of 59 males and 193 females with a mean age of 46, range 15 to 85 years, included 32 PM/DM patients, 15 with MCTD, 10 cutaneous lupus cases, 83 diagnosed with SLE, 36 with SS, and 76 SSc patients.
The antinuclear antibodies (ANA) were detected using NOVA Lite HEp-2 ANA kit (Inova Diagnostics, Inc.; San Diego, CA, USA). The slide processing was carried out on Inova’s QUANTA-Lyser 2 and read using their NOVA View digital Indirect Immunofluorescence (IIF) microscope software. NOVA View is an automated fluorescent microscope programmed to acquire, archive and manage digital images of fluorescent stained slides. The system encloses an Olympus 1×81 inverted fluorescent microscope (Olympus Belgium N.V.; Aartselaar, Belgium). Likelihood ratios were calculated for fluorescence intensity result intervals.
The investigators found a significant correlation between end-point titer and fluorescence intensity. Likelihood ratios for a systemic rheumatic disease increased with increasing fluorescence intensity. The likelihood ratio for a systemic rheumatic disease was 0.06, 0.18, 0.51, 5.3, and 37.5 for a fluorescence intensity of equal to or greater than 66, 67 to 150, 151 to 300, 301 to 1,000, greater than 1,000, respectively. A range of 31% to 37% of the patients with Sjögren’s syndrome, systemic sclerosis or systemic lupus erythematosus had fluorescence intensities greater than 1,000.
The authors concluded that estimations of fluorescence intensity by automated antinuclear antibody analysis offers clinically useful information. Likelihood ratios based on fluorescence intensity test result intervals aid with the interpretation of automated antinuclear antibody analysis and allow value-added reporting. The study was published on April 1, 2014, in the journal Clinical Chemistry and Laboratory Medicine.
Related Links:
University Hospitals Leuven
Inova Diagnostics
Olympus Belgium NV














