Immunoassay Detects Antibodies to American Trypanosomiasis
By LabMedica International staff writers
Posted on 11 Jan 2011
A fully automated immunoassay can determine the presence of antibodies to Trypanosoma cruzi, the causative agent of Chagas disease, in human serum and plasma.Posted on 11 Jan 2011
The quantitative two-step chemiluminescent microparticle immunoassay (CMIA) has been compared to an enzyme linked immunoassays (ELISA) for specificity and sensitivity. In the first step, sample, assay diluent, and paramagnetic microparticles co-coated with four chimeric T. cruzi recombinant antigens, are combined. A murine acridinium-labeled antihuman immunoglobulin (IgG) conjugate is added. After another wash cycle, pre-trigger and trigger solutions are added to the reaction mixture.
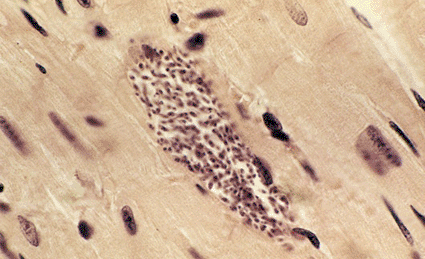
Image: Light micrograph of a section through a cluster of Trypanosoma cruzi protozoans (purple, center) in the heart muscle of a patient suffering from Chagas disease (Photo courtesy of Sinclair Stammers / Science Photo Library).
The ARCHITECT Chagas prototype assay (Abbott GmbH & Co.; Wiesbaden-Delkenheim, Germany) was tested on total of 10, 284 specimens to determine the specificity and sensitivity of the assay. The tests were done in Abbott's laboratory in Germany and in external sites in Guatemala and Brazil.
Of 10,284 specimens tested, 9,629 had a final interpretation of T. cruzi antibody-negative based on the testing algorithm used, whereas 655 specimens were determined to be T. cruzi antibody-positive. In comparison with the ELISA cruzi assay (bioMérieux; Marcy l'Etoile, France; www.biomerieux.com), the ARCHITECT assay demonstrated superior specificity (99.99% vs. 99.93%) and sensitivity (99.85% vs. 98.38%), along with excellent precision. This suggests that this CMIA has the potential to serve as single assay to determine the T. cruzi status of a given blood unit or diagnostic specimen on a fully automated instrument platform.
The authors of the study concluded that the utility of the fully automated ARCHITECT Chagas assay can be used in the diagnostic setting and for blood donor testing in endemic as well as nonendemic regions. The assay, once it is commercially available, might be used as a single test for routine testing in high-prevalence areas such as South and Central America, which would enable an economic testing strategy being highly advantageous for the target countries. Chagas disease or American Trypanosomiasis represents the third largest tropical disease burden worldwide, after malaria and schistosomiasis. The study was published in January 2011, in Diagnostic Microbiology and Infectious Disease.
Related Links:
bioMérieux
Abbott














