Emerging Biomarkers Advance Early Detection of MASLD and Liver Cancer Risk
Posted on 12 May 2026
Metabolic dysfunction-associated steatotic liver disease (MASLD) affects about 30% of people worldwide and can advance to metabolic dysfunction-associated steatohepatitis (MASH), fibrosis, cirrhosis, and hepatocellular carcinoma (HCC). Reliance on liver biopsy and traditional serum markers hampers early, accurate detection across this spectrum. As incidence rises, laboratories need validated noninvasive tools and clearer therapeutic strategies.
A new review in Science Bulletin outlines emerging diagnostic approaches, notably multi-omics panels that integrate genetic, metabolic, and microbiome markers. These panels are presented as noninvasive alternatives intended to replace or reduce reliance on liver biopsy, with the goal of earlier and more precise detection. The review notes diagnostic performance that exceeds traditional assessments used for staging MASH and fibrosis.
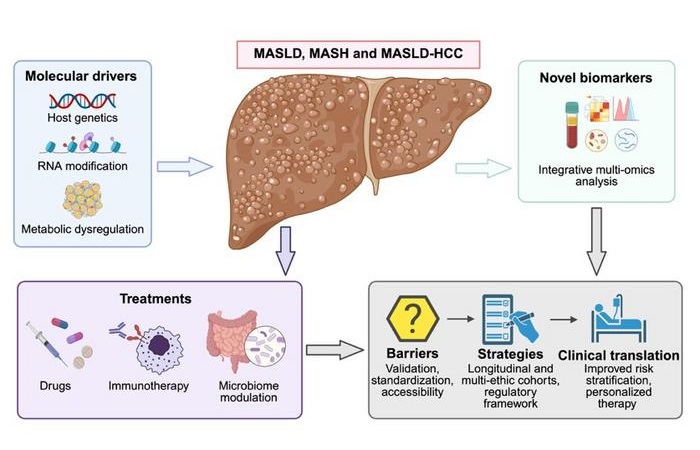
According to the review, gut microbiome signatures—comprising specific bacterial taxa and their metabolites—can differentiate disease stages and flag individuals at risk. Molecular biomarkers, including microRNAs and serum proteins, are highlighted for early HCC surveillance, with performance described as superior to alpha-fetoprotein. Integrating such multi-omics readouts could improve risk stratification and facilitate objective monitoring of treatment response in routine practice.
Therapeutically, the review notes that recent U.S. Food and Drug Administration (FDA) approvals of resmetirom and semaglutide represent a milestone, providing the first pharmacological options specifically for patients with MASH and moderate-to-advanced fibrosis. A broader pipeline of phase 3 agents targeting metabolic pathways aims to reduce hepatic steatosis, quell inflammation, and reverse fibrosis. For MASLD-associated HCC, however, diminished responses to immune checkpoint inhibitors are attributed to an immunosuppressive tumor microenvironment, underscoring the need for tailored combination regimens.
The authors also identify key barriers to clinical translation, including limited analytical and clinical validation across diverse, multiethnic cohorts and the inherent heterogeneity of MASLD, which complicates the use of uniform diagnostic cutoffs. The review calls for large, prospective longitudinal studies to confirm prognostic value and cost-effectiveness, along with standardized analytical workflows and clear regulatory frameworks. It also supports integrating validated noninvasive biomarkers into electronic health records to enable systematic screening and real-world monitoring.