Point-of-Care System Evaluated for Measuring Cardiac Troponin I
By LabMedica International staff writers
Posted on 13 Dec 2016
Measurement of cardiac Troponin-I or Troponin-T (cTnI, cTnT) concentration in blood is required for assessment of patients suspected of Non ST-segment elevation acute myocardial infarction (NSTE-AMI) to support or exclude a diagnosis of acute myocardial infarction (AMI).Posted on 13 Dec 2016
The measurement of cTnI at the point of care with a short turnaround time (TAT) has the potential to improve patients flow in the emergency department (ED), enabling rapid clinical decision making. A patient blood sample can be withdrawn and directly tested by a doctor, a nurse or a paramedic to provide cTnI concentration during clinical examination.
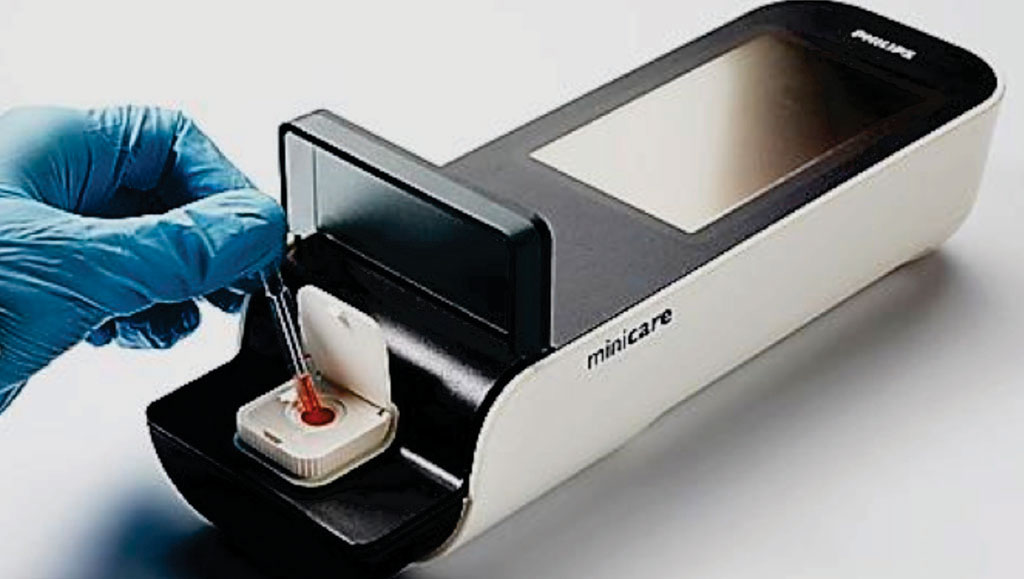
Image: The Minicare I-20 system point-of-care blood test for the rapid diagnosis of a heart attack (Photo courtesy of Philips Healthcare).
A European team of clinicians working with Philips Handheld Diagnostics (Eindhoven, The Netherlands) collected from patients at four European hospitals three sample types: capillary whole blood from finger stick, Li-heparin whole blood and Li-heparin plasma. Samples were analyzed by trained users, nurses and research assistants within two hours after the blood was drawn. Frozen samples were sent to the core laboratory at Philips on dry ice for parallel testing on the Beckman Coulter AccuTnI + 3 assay (Beckman Coulter Life Sciences, Indianapolis IN, USA) and the Philips Minicare cTnI for the method comparison study.
The Philips Minicare cTnI consists of a handheld instrument and plastic disposable cartridge. The system makes use of the Philips Magnotech technology, which is based on the precisely controlled motion of magnetic particles (beads) in a small sample volume (typically 30 μL). The same magnetic particles also serve as labels that are detected using frustrated total internal reflection (FTIR) imaging. The Minicare cTnI assay is a homogeneous sandwich immunoassay. The traditional liquid manipulation steps of an immunoassay have been replaced by magnetically controlled movements of magnetic nanoparticles within a stationary liquid.
The sample type comparison study between capillary blood, Li-heparin whole blood and Li-heparin plasma samples demonstrated very high correlation coefficients between 0.99 and 1.00, and the method comparison between Minicare cTnI and Beckman Coulter Access, AccuTnI + 3 demonstrated a correlation coefficient of 0.973. No high-dose hook effect was found for samples up to and including a cTnI concentration of 2,000,000 ng/L. The limit of the blank, limit of detection and limit of quantitation at 20% coefficient of variation (CV) were determined to be 8.5 ng/L, 18 ng/L and 38 ng/L respectively. Total CV was found to be from 7.3% to 12% for cTnI concentrations between 109.6 ng/L and 6,135.4 ng/L.
The authors concluded that the Minicare cTnI assay is a sensitive, fast and precise test for determination of cTnI that can be used as an aid in the diagnosis of AMI in a near-patient setting on capillary or Li-heparin venous whole blood sample. This offers for the Minicare cTnI test the potential for good clinical performance in the diagnosis of AMI. The study was published online on November 12, 2016, in the journal Clinical Biochemistry.
Related Links:
Philips Healthcare
Beckman Coulter Life Sciences






.jpg)