Electrical Immunosensor Detects Acute Myocardial Infarction
By LabMedica International staff writers
Posted on 25 Aug 2016
Heart disease and especially acute myocardial infarction (AMI) are the leading causes of death for both men and women and therefore, a fast and reliable diagnosis of heart attack or cardiac episode are urgently needed.Posted on 25 Aug 2016
The most commonly used biomarkers are creatine kinase-MB, myoglobin, cardiac troponin T, and cardiac troponin I (cTnI), which is a subunit of the troponin complex found in cardiac muscle and is a highly specific and sensitive biomarker for the clinical diagnosis of AMI.
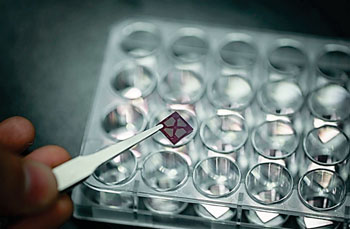
Image: The core material used for the new immunosensor that detects proteins in the blood stream following a heart attack, providing results in just one minute (Photo courtesy of Ulsan National Institute of Science and Technology).
Biomedical engineers at Ulsan National Institute of Science and Technology (South Korea) developed a new sensor for early detection of heart attack in humans. The apparatus comprises of rapid, label-free, and highly sensitive single-walled carbon nanotube (SWCNT) electrical immunosensor, featuring two pairs of electrodes. The system works by measuring the level of cardiac troponin I (cTnI), a protein that is excreted by the heart muscle into the blood following a heart attack.
The two concentration electrodes were imbedded between upper and lower dielectric layers, facing each other, underneath the -COOH-functionalized SWCNT channels deposited between the detection electrodes. Therefore, the gap between these imbedded concentration electrodes can be reduced to maximize the electric field intensity for dielectrophoresis (DEP)-mediated concentration of cTnI, thereby greatly reducing the detection time to one minute and enhancing the limit of detection (0.7–0.8 pg/mL). Fluorescence images were acquired using an Eclipse 80i epifluorescence microscope (Nikon, Tokyo, Japan).
The authors concluded that their immunosensor demonstrated high selectivity for cTnI over myoglobin, Tris-Borate-EDTA (TBE), and human serum. Therefore, the novel immunosensor presented holds considerable potential for use as a platform for sensing distinct types of proteins, along with the feasibility of miniaturization and integration for biomedical diagnosis. The study was published in the August 2016 issue of the journal Biosensors & Bioelectronics.
Related Links:
Ulsan National Institute of Science and Technology
Nikon