Blood Immune 'Fingerprint' Predicts Side Effects of New Alzheimer's Drug
Posted on 16 Mar 2026
New antibody therapies for Alzheimer’s disease have shown promise in slowing disease progression, but their broader use has been limited by a common side effect known as amyloid-related imaging abnormalities (ARIA). ARIA can appear as brain swelling or small hemorrhages on MRI scans and remains difficult to predict before treatment begins. Researchers have now identified a blood-based immune signature that may help predict which patients are more likely to develop this complication.
Researchers at the University of Kentucky (Lexington, KY, USA) examined blood samples from patients receiving lecanemab, an anti-amyloid antibody therapy approved by the U.S. Food and Drug Administration to slow Alzheimer’s disease progression. The investigators focused on identifying biological signals associated with ARIA in patients treated for Alzheimer’s. Using advanced genetic and metabolic profiling, the researchers analyzed blood samples from patients undergoing lecanemab treatment. They discovered that individuals who developed ARIA showed an expansion of a specific subset of T cells in the bloodstream.
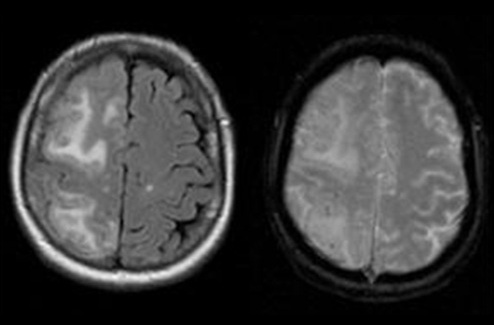
These immune cells displayed increased metabolic activity and molecular characteristics suggesting they were primed to interact with the vascular system. The coordinated immune pattern formed a distinct biological signature that differentiated patients who developed ARIA from those who did not. Importantly, the immune signature was detectable through a simple blood test, offering a potential noninvasive method to identify patients at higher risk for treatment-related complications.
Previously, the primary known risk factor for ARIA was the APOE ε4 genetic variant, which increases susceptibility to Alzheimer’s disease and certain treatment side effects. The new findings, published in Nature Communications, provide insight into the biological mechanisms underlying ARIA and suggest that immune system activity may play an important role. If validated in larger patient groups, the immune signature identified in the study could form the basis of a predictive screening test for ARIA risk.
Such a tool could allow clinicians to personalize treatment strategies by adjusting monitoring frequency, dosing schedules or supportive interventions for patients at higher risk. Researchers emphasize that further studies are needed before the approach can be used clinically, but the findings represent an important step toward improving the safety of emerging Alzheimer’s therapies.
“We asked a simple question: do people who develop ARIA show a different immune ‘fingerprint’ in their blood than those who don’t? The answer appears to be yes,” said Josh Morganti, PhD, assistant professor of neuroscience in the UK College of Medicine and researcher at the University of Kentucky’s Sanders-Brown Center on Aging.
Related Links:
University of Kentucky