Experimental Drug Slows Lung Cancer Growth by Blocking Protein Glycosylation
By LabMedica International staff writers
Posted on 03 Nov 2016
An interesting new experimental anti-cancer drug slows growth of certain lung tumor cells by preventing the glycosylation of critical cell surface receptor proteins.Posted on 03 Nov 2016
Asparagine (N)-linked glycosylation is a protein modification process that is critical for glycoprotein folding, stability, and cellular localization. To identify small molecules that would inhibit new targets in this biosynthetic pathway, investigators at Yale University (New Haven, CT, USA) and colleagues at several other institutions initiated a cell-based high-throughput screen and lead-compound-optimization campaign. During this campaign the researchers screened more than 350,000 chemical compounds while searching for those that could partially disrupt glycosylation. The result was the small molecule cell-permeable inhibitor NGI-1.
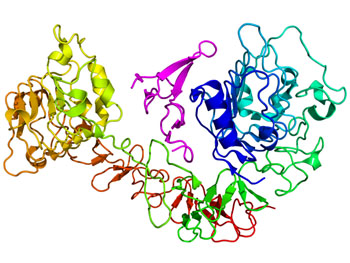
Image: The experimental drug NGI-1 slows cancer growth by blocking glycosylation of the epidermal growth factor receptor (EGFR), which is shown in the above diagram (Photo courtesy of Wikimedia Commons).
The investigators reported in the October 3, 2016, online edition of the journal Nature Chemical Biology that NGI-1 targeted oligosaccharyltransferase (OST), a hetero-oligomeric enzyme that exists in multiple isoforms and transfers oligosaccharides to recipient proteins.
In non-small-cell lung cancer (NSCLC) cells, NGI-1 blocked cell-surface localization and signaling of the epidermal growth factor receptor (EGFR) glycoprotein, but selectively arrested proliferation in only those cell lines that were dependent on EGFR (or fibroblast growth factor, FGFR) for survival. In these cell lines, OST inhibition caused cell-cycle arrest accompanied by cell morphology changes that were hallmarks of senescence.
“This is important to cancer research because what we are looking for are therapies that do not have a lot of effect on normal cells but do have a lot of effect on tumor cells, and this falls into that category,” said senior author Dr. Joseph Contessa, associate professor of therapeutic radiology and of pharmacology at Yale University. “We have therapies, and they are good therapies, but they are not enough. We need to take the next step.”
Related Links:
Yale University