Loss-of-Function Mutation Causes ALS in Mouse Model
By LabMedica International staff writers
Posted on 25 Jul 2016
Neurological disease researchers working with a mouse model of amyotrophic lateral sclerosis (ALS) have shown that the condition is critically linked to a loss-of-function mutation in the C9ORF72 (chromosome 9 open reading frame 72) gene.Posted on 25 Jul 2016
C9ORF72 mutations have been found in a significant fraction of patients suffering from ALS and frontotemporal dementia (FTD), yet the function of the C9ORF72 gene product remains poorly understood.
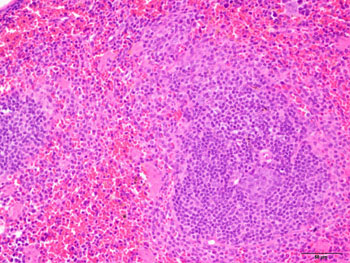
Image: In the mouse spleen, lymphoid tissue (purple) is responsible for launching an immune response to blood-born antigens, while red pulp (pink) filters the blood. Mutations in the C9ORF72 gene, the most common mutation found in ALS patients, can inflame lymphoid tissue and contribute to immune system dysfunction (Photo courtesy of Dr. Dan Mordes, Eggan Laboratory, Harvard Stem Cell Institute).
In a study published in the July 14, 2016, online edition of the journal Science Translational Medicine investigators at Harvard University (Cambridge, MA, USA) and the Massachusetts Institute of Technology (Cambridge, MA, USA) described results of experiments performed with an ALS mouse model.
They reported that mice carrying loss-of-function mutations in C9ORF72 developed splenomegaly, neutrophilia, thrombocytopenia, increased expression of inflammatory cytokines, and severe autoimmunity, ultimately leading to a high mortality rate. Transplantation of mutant mouse bone marrow into wild-type recipients was sufficient to create the phenotypes observed in the mutant animals, including autoimmunity and premature mortality. Reciprocally, transplantation of wild-type mouse bone marrow into mutant mice improved their phenotype.
Knockout mice – genetically engineered to lack the C9ORF72 gene – that received a bone marrow transplant from normal animals lived on average 43 days longer, weighed more, and had a greater number of blood platelets throughout their lifespan than did untreated knockout mice.
"The point of our paper was to determine the function of this gene and what it normally helps to do in the body," said senior author Dr. Kevin Eggan, professor of stem cell and regenerative biology at Harvard University. "The mice seemed to get better, but the bone marrow transplant did not wholly rescue the animals, suggesting that there may also be functions of C9ORF72 in other organs."
Related Links:
Harvard University
Massachusetts Institute of Technology