Selective Ribosome-Targeting Drug with Low Ototoxicity Could Provide Safer Treatment Option for Leishmaniasis
By LabMedica International staff writers
Posted on 03 Sep 2015
In a multidisciplinary biological study, researchers have shed new light on the mechanism of action of a potent drug for leishmaniasis, the natural aminoglycoside paromomycin (PAR), and have found that a new synthetic derivative of PAR may provide a far lower risk of irreversible hearing loss side-effects, probably via its more selective targeting of cytosolic (vs. mitochondrial) ribosomes.Posted on 03 Sep 2015
The limited arsenal of drugs currently available for leishmaniasis therapy, coupled with the rather fast emergence of parasite resistance, presents a dire public health concern. A promising approach is the delineation of new cellular drug-targets through mechanistic studies. Data from structural, biochemical, and pathological experiments are valuable for rational design and development of new potential therapeutic agents. The leishmanial ribosome is one potential target of antileishmanial therapeutics, especially for derivatives belonging to the important aminoglycosides (AG) family.
In the new study, a team of researchers, led by Timor Baasov, professor at the Technion - Israel Institute of Technology (Haifa, Israel), investigated the structural basis for the targeting of leishmanial ribosomes by AGs and performed comparative testing of the potential clinical effectiveness and side-effects of new (semi-) synthetic derivatives of PAR. The study was a collaboration of researchers from the Technion, the Weizmann Institute of Science (Rehovot, Israel), the Hebrew University-Hadassah Medical School (Jerusalem, Israel), and the University of Michigan Medical School (Ann Arbor, MI, USA).
PAR, a broad-spectrum antibiotic, is highly efficient in treating life-threatening visceral leishmaniasis (VL). In 2007, the World Health Organization (WHO) listed PAR injection for VL as an essential medication due to its high potency, accessibility, and affordability in highly endemic countries. Also, a new combination therapy of PAR given with liposomal amphotericin B has shown potential to be short, safe, and effective, and would help prevent development of drug resistance and lower cost compared to treatment with liposomal amphotericin B alone.
However, PAR can have severe ototoxic side-effects. In mild cutaneous leishmaniasis (CL), this toxicity is less problematic as single lesions are generally treated topically, however in CL where multiple lesions are present, or in mucocutaneous leishmaniasis and VL where PAR or other AGs are given systemically, toxicity often becomes a serious problem.
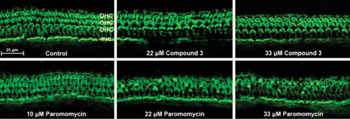
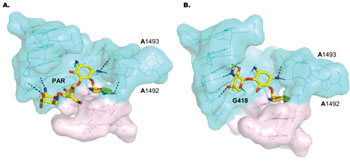
While much focus has been given to exploration of PAR activities in bacteria, its mechanism of action in Leishmania has received relatively little attention. In the new study, the researchers present, for the first time, X-ray crystallography results of PAR bound in complex with a ribosomal RNA (rRNA) model mimicking two leishmanial cytosolic ribosomal binding sites (ribosomal “A-sites”). They also evaluated inhibitory actions on leishmanial growth and ribosome function, as well as effects on mammalian auditory sensory cells, by comparing structurally related AGs. The results provided insights into the structural elements important for AG inhibitory activities and selectivity for leishmanial cytosolic ribosomes, and highlighted a novel synthetic derivative of PAR, compound-3, as a prospective candidate for effective treatment. In preliminary in vivo experiments in a guinea pig model, compound-3 exhibited low auditory toxicity compared to PAR, and so could eliminate a major drawback that limits clinical use of PAR and other currently available AGs.
The study, by Shalev M, et al., was reported online, ahead of print, August 11, 2015, in the journal Nucleic Acids Research.
Related Links:
Technion - Israel Institute of Technology