Researchers Describe Novel Methods for Disabling Bacterial Proteases
By LabMedica International staff writers
Posted on 27 Feb 2014
A paper described previously unrecognized mechanisms that can be used to permanently deactivate critical bacteriological proteases.Posted on 27 Feb 2014
Proteases are responsible for the pathogenic effects of many kinds of bacteria, and considerable research effort is involved in developing effective ways of inhibiting their action.
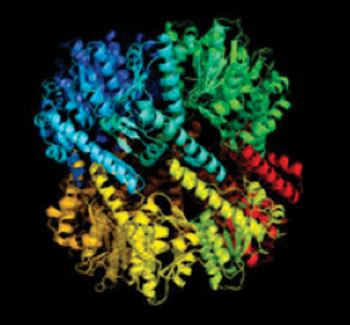
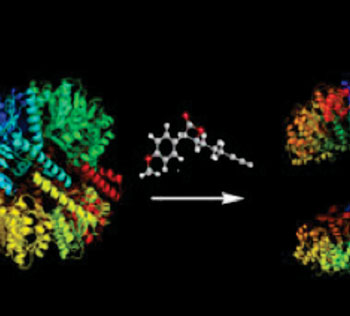
Investigators at the Technische Universitaet Muenchen (Munich, Germany) recently described novel approaches for silencing the activity of bacterial ClpP protease. In the January 29, 2014, issue of the Journal of the American Chemical Society they presented a novel mechanism of protease inhibition that relied on active-site-directed small molecules that disassembled the protease complex. They showed the applicability of this mechanism within the ClpP protease family, whose members are tetradecameric serine proteases and serve as regulators of several cellular processes, including homeostasis and virulence.
In addition, they reported the selective beta-sultam-induced dehydroalanine formation of the active site serine. This reaction proceeded through sulfonylation and subsequent elimination, thereby obliterating the catalytic charge relay system. The identity of the dehydroalanine was confirmed by mass spectrometry and crystallography. Activity-based protein profiling experiments suggested the formation of a dehydroalanine moiety in living Staphylococcus aureus cells upon beta-sultam treatment.
The mechanisms described here point towards the possibility of developing protease inhibitors that do not rely on complete blocking of the enzymes' catalytic or binding sites.
“ClpP inhibitors used in the past have one decisive disadvantage,” said senior author Dr. Stephan Sieber, professor of organic chemistry at the Technische Universitaet Muenchen. “They do not permanently disarm the proteins, but only work for a few hours. On top of that, to be effective they must attack all active centers of the protein.”
Related Links:
Technische Universitaet Muenchen