HemoSonics Demonstrates Breakthrough in Point-Of-Care Viscoelastic Hemostasis Testing
Posted on 01 Aug 2024
At ADLM 2024, HemoSonics (Durham, NC, USA) is demonstrating its Quantra Hemostasis System which has the broadest range of FDA-cleared indications for use in its category and is cleared for use with arterial and venous blood samples in cardiac, trauma, liver, and major orthopedic surgery patients.
HemoSonics' Quantra Hemostasis System consists of the Quantra Hemostasis Analyzer with QPlus and QStat Cartridges, which enable clinicians to deliver patient-centered decisions on bleeding management that improve care and optimize blood product usage. The Quantra System is FDA-cleared for use in point-of-care settings such as operating rooms, intensive care units, and in hospital laboratories, enabling hospitals to standardize use institution-wide with greater operational efficiency. The Quantra System allows healthcare professionals to make individualized and evidence-based decisions on how to manage a bleeding patient and give a patient only medically appropriate therapy – an approach that supports global initiatives to reduce inappropriate transfusions of blood products.
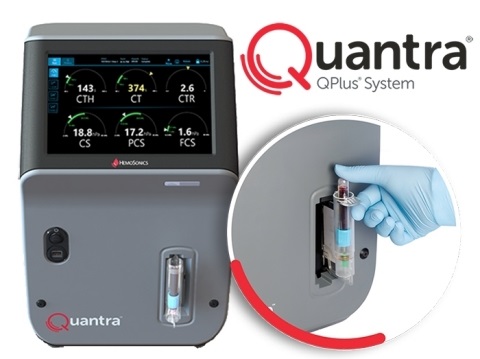
The Quantra System requires minimal resources to maintain, is easy to operate and interpret, and provides fast, comprehensive whole-blood coagulation analysis at the point of care or in laboratory-based settings, typically in less than 15 minutes. A recent study showed that the implementation of the Quantra Hemostasis Analyzer with the QPlus Cartridge reduced utilization of blood components by up to 90%, allowing greater availability for those patients truly in need. In addition, the cost of blood products decreased by 41%. HemoSonics' QStat Cartridge used with the Quantra Hemostasis Analyzer first received 510(k) clearance from the U.S. FDA in 2022 for use in venous whole blood samples. In April this year, HemoSonics received Special 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the expanded use of arterial blood samples with its Quantra QStat Cartridge.
Related Links:
HemoSonics




.jpg)








.jpg)
