Roche Completes Acquisition of TIB Molbiol Group
|
By LabMedica International staff writers Posted on 02 Dec 2021 |

Roche (Basel, Switzerland) has completed its share purchase agreement to acquire 100% of the outstanding shares of TIB Molbiol Group (Berlin, Germany).
TIB Molbiol will continue to operate as a subsidiary within the Diagnostics division. Roche and TIB Molbiol will build on their capabilities for the rapid development of assays for emerging pathogens and potential health threats, such as infectious diseases. The two companies have collaborated for more than 20 years to rapidly address critical healthcare needs including biological threats, such as SARS, anthrax, avian influenza virus H5N1, MERS, the novel influenza virus H1N1 swine, Ebola virus, Zika virus and most recently, SARS-CoV-2 virus and its variants. For example, in 2001 with anthrax and 2003 with SARS-CoV1, TIB Molbiol demonstrated their ability to develop PCR assays for the detection of new pathogens within days.
With over 45 CE-IVD assays and more than a 100 research-use-only assays joining the Roche family, the companies will jointly accelerate access for patients around the world to even more clinically important diagnostic tests. TIB Molbiol will further expand Roche’s molecular diagnostics solutions, especially for the flexible open platforms LightCycler PCR and MagNA Pure sample preparation systems.
“I am very pleased that together with TIB Molbiol, we can significantly improve access for patients to a broader spectrum of diagnostic tests - from high volume routine tests to the diagnosis of rare diseases. TIB Molbiol will continue to be an innovation engine and frontrunner in our common fight against infectious diseases, such as SARS-CoV-2,” said Thomas Schinecker, CEO Roche Diagnostics. “TIB Molbiol has helped to shape the future of PCR infectious disease testing, and will continue to drive cutting-edge research so we can bring better healthcare outcomes to patients faster.”
Related Links:
Roche
TIB Molbiol
Latest Industry News
- ECCMID Congress Name Changes to ESCMID Global
- Bosch and Randox Partner to Make Strategic Investment in Vivalytic Analysis Platform
- Siemens to Close Fast Track Diagnostics Business
- Beckman Coulter and Fujirebio Expand Partnership on Neurodegenerative Disease Diagnostics
- Sysmex and Hitachi Collaborate on Development of New Genetic Testing Systems
- Sysmex and CellaVision Expand Collaboration to Advance Hematology Solutions
- BD and Techcyte Collaborate on AI-Based Digital Cervical Cytology System for Pap Testing
- Medlab Middle East 2024 to Address Transformative Potential of Artificial Intelligence
- Seegene and Microsoft Collaborate to Realize a World Free from All Diseases and Future Pandemics
- Medlab Middle East 2024 to Highlight Importance of Sustainability in Laboratories
- Fujirebio and Agappe Collaborate on CLIA-Based Immunoassay
- Medlab Middle East 2024 to Highlight Groundbreaking NextGen Medicine
- bioMérieux Acquires Software Company LUMED to Support Fight against Antimicrobial Resistance
- Roche Acquires LumiraDx's Point of Care Technology for USD 295 Million
- Bruker Acquires IVD Manufacturer ELITech Group and Cell Imaging Company Phasefocus
- AMP Publishes Best Practice Guidance for Slice Testing Approach in Diagnostics
Channels
Clinical Chemistry
view channel
3D Printed Point-Of-Care Mass Spectrometer Outperforms State-Of-The-Art Models
Mass spectrometry is a precise technique for identifying the chemical components of a sample and has significant potential for monitoring chronic illness health states, such as measuring hormone levels... Read more.jpg)
POC Biomedical Test Spins Water Droplet Using Sound Waves for Cancer Detection
Exosomes, tiny cellular bioparticles carrying a specific set of proteins, lipids, and genetic materials, play a crucial role in cell communication and hold promise for non-invasive diagnostics.... Read more
Highly Reliable Cell-Based Assay Enables Accurate Diagnosis of Endocrine Diseases
The conventional methods for measuring free cortisol, the body's stress hormone, from blood or saliva are quite demanding and require sample processing. The most common method, therefore, involves collecting... Read moreMolecular Diagnostics
view channel
Blood Samples Enhance B-Cell Lymphoma Diagnostics and Prognosis
B-cell lymphoma is the predominant form of cancer affecting the lymphatic system, with about 30% of patients with aggressive forms of this disease experiencing relapse. Currently, the disease’s risk assessment... Read more
Blood Test Predicts Knee Osteoarthritis Eight Years Before Signs Appears On X-Rays
Osteoarthritis (OA) is the most prevalent form of arthritis, impacting millions worldwide and resulting in significant economic and social costs. Although no cure exists currently, the effectiveness of... Read more
Blood Test Accurately Predicts Lung Cancer Risk and Reduces Need for Scans
Lung cancer is extremely hard to detect early due to the limitations of current screening technologies, which are costly, sometimes inaccurate, and less commonly endorsed by healthcare professionals compared... Read more
Unique Autoantibody Signature to Help Diagnose Multiple Sclerosis Years before Symptom Onset
Autoimmune diseases such as multiple sclerosis (MS) are thought to occur partly due to unusual immune responses to common infections. Early MS symptoms, including dizziness, spasms, and fatigue, often... Read moreHematology
view channel
Next Generation Instrument Screens for Hemoglobin Disorders in Newborns
Hemoglobinopathies, the most widespread inherited conditions globally, affect about 7% of the population as carriers, with 2.7% of newborns being born with these conditions. The spectrum of clinical manifestations... Read more
First 4-in-1 Nucleic Acid Test for Arbovirus Screening to Reduce Risk of Transfusion-Transmitted Infections
Arboviruses represent an emerging global health threat, exacerbated by climate change and increased international travel that is facilitating their spread across new regions. Chikungunya, dengue, West... Read more
POC Finger-Prick Blood Test Determines Risk of Neutropenic Sepsis in Patients Undergoing Chemotherapy
Neutropenia, a decrease in neutrophils (a type of white blood cell crucial for fighting infections), is a frequent side effect of certain cancer treatments. This condition elevates the risk of infections,... Read more
First Affordable and Rapid Test for Beta Thalassemia Demonstrates 99% Diagnostic Accuracy
Hemoglobin disorders rank as some of the most prevalent monogenic diseases globally. Among various hemoglobin disorders, beta thalassemia, a hereditary blood disorder, affects about 1.5% of the world's... Read moreImmunology
view channel
Diagnostic Blood Test for Cellular Rejection after Organ Transplant Could Replace Surgical Biopsies
Transplanted organs constantly face the risk of being rejected by the recipient's immune system which differentiates self from non-self using T cells and B cells. T cells are commonly associated with acute... Read more
AI Tool Precisely Matches Cancer Drugs to Patients Using Information from Each Tumor Cell
Current strategies for matching cancer patients with specific treatments often depend on bulk sequencing of tumor DNA and RNA, which provides an average profile from all cells within a tumor sample.... Read more
Genetic Testing Combined With Personalized Drug Screening On Tumor Samples to Revolutionize Cancer Treatment
Cancer treatment typically adheres to a standard of care—established, statistically validated regimens that are effective for the majority of patients. However, the disease’s inherent variability means... Read moreMicrobiology
view channel
Clinical Decision Support Software a Game-Changer in Antimicrobial Resistance Battle
Antimicrobial resistance (AMR) is a serious global public health concern that claims millions of lives every year. It primarily results from the inappropriate and excessive use of antibiotics, which reduces... Read more
New CE-Marked Hepatitis Assays to Help Diagnose Infections Earlier
According to the World Health Organization (WHO), an estimated 354 million individuals globally are afflicted with chronic hepatitis B or C. These viruses are the leading causes of liver cirrhosis, liver... Read more
1 Hour, Direct-From-Blood Multiplex PCR Test Identifies 95% of Sepsis-Causing Pathogens
Sepsis contributes to one in every three hospital deaths in the US, and globally, septic shock carries a mortality rate of 30-40%. Diagnosing sepsis early is challenging due to its non-specific symptoms... Read morePathology
view channel
First of Its Kind Universal Tool to Revolutionize Sample Collection for Diagnostic Tests
The COVID pandemic has dramatically reshaped the perception of diagnostics. Post the pandemic, a groundbreaking device that combines sample collection and processing into a single, easy-to-use disposable... Read moreAI-Powered Digital Imaging System to Revolutionize Cancer Diagnosis
The process of biopsy is important for confirming the presence of cancer. In the conventional histopathology technique, tissue is excised, sliced, stained, mounted on slides, and examined under a microscope... Read more
New Mycobacterium Tuberculosis Panel to Support Real-Time Surveillance and Combat Antimicrobial Resistance
Tuberculosis (TB), the leading cause of death from an infectious disease globally, is a contagious bacterial infection that primarily spreads through the coughing of patients with active pulmonary TB.... Read moreTechnology
view channel
New Diagnostic System Achieves PCR Testing Accuracy
While PCR tests are the gold standard of accuracy for virology testing, they come with limitations such as complexity, the need for skilled lab operators, and longer result times. They also require complex... Read more
DNA Biosensor Enables Early Diagnosis of Cervical Cancer
Molybdenum disulfide (MoS2), recognized for its potential to form two-dimensional nanosheets like graphene, is a material that's increasingly catching the eye of the scientific community.... Read more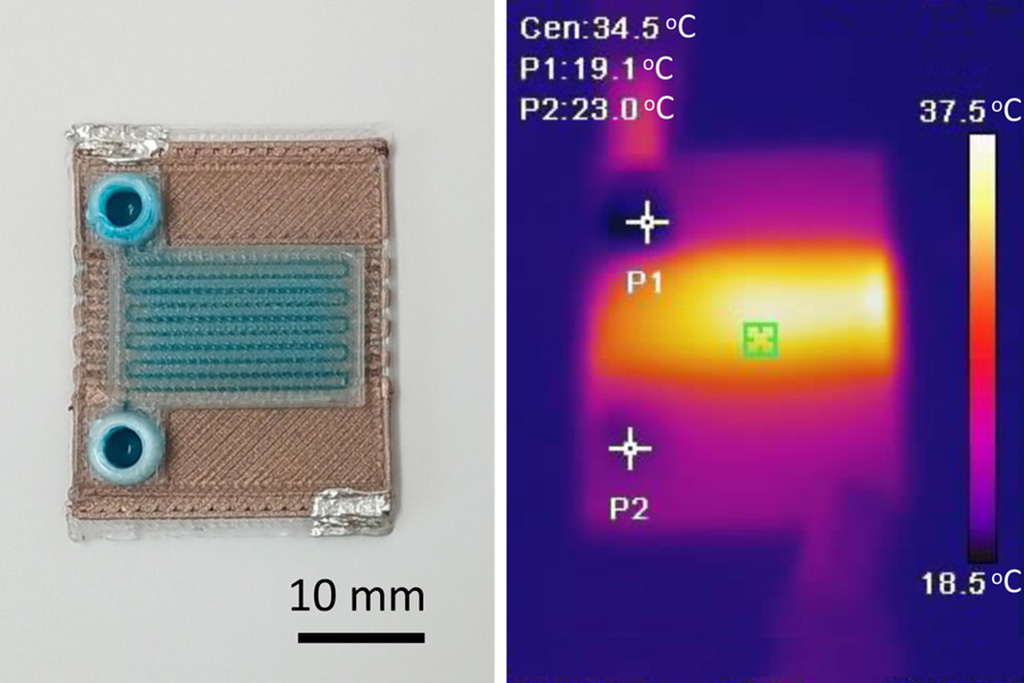
Self-Heating Microfluidic Devices Can Detect Diseases in Tiny Blood or Fluid Samples
Microfluidics, which are miniature devices that control the flow of liquids and facilitate chemical reactions, play a key role in disease detection from small samples of blood or other fluids.... Read more