Genome Analysis Shows Meiosis Gene Mutation Causes Primary Ovarian Insufficiency
|
By LabMedica International staff writers Posted on 28 Dec 2014 |
![Image: In meiosis, the chromosomes duplicate (during interphase) and homologous chromosomes exchange genetic information (chromosomal crossover) before a first division, called meiosis I. The daughter cells divide again in meiosis II, splitting up sister chromatids to form haploid gametes. Male and female gametes fuse during fertilization, creating a diploid cell with a complete set of paired chromosomes (Photo courtesy of the [US] National Institutes of Health). Image: In meiosis, the chromosomes duplicate (during interphase) and homologous chromosomes exchange genetic information (chromosomal crossover) before a first division, called meiosis I. The daughter cells divide again in meiosis II, splitting up sister chromatids to form haploid gametes. Male and female gametes fuse during fertilization, creating a diploid cell with a complete set of paired chromosomes (Photo courtesy of the [US] National Institutes of Health).](https://globetechcdn.com/mobile_labmedica/images/stories/articles/article_images/2014-12-28/GMS-331.jpg)
Image: In meiosis, the chromosomes duplicate (during interphase) and homologous chromosomes exchange genetic information (chromosomal crossover) before a first division, called meiosis I. The daughter cells divide again in meiosis II, splitting up sister chromatids to form haploid gametes. Male and female gametes fuse during fertilization, creating a diploid cell with a complete set of paired chromosomes (Photo courtesy of the [US] National Institutes of Health).
A mutation in a gene linked to the process of meiosis was found in two sisters diagnosed with primary ovarian insufficiency (POI), a syndrome that prevented them from reaching sexual maturity.
Mammals and many other organisms achieve reduction and recombination through a process called meiosis, which is recognizable by the presence of a distinctive structure—the synaptonemal complex (SCS), —that links the chromosomes together and is essential for meiosis to complete. The protein encoded by the SYCE1 gene (synaptonemal complex central element 1) may have a role in the synaptonemal complex assembly, stabilization, and recombination. Genes involved in essential steps in chromosome synapsis and recombinations during meiosis, such as SYCE1, have been shown to cause POI in animal models but not in humans.
Investigators at Tel Aviv University (Israel) worked with two daughters of consanguineous parents (first cousins) from a 13-member Israeli-Arab family who had been diagnosed with POI. Genotyping was performed in the patients, their parents, four unaffected siblings, and a group of 90 ethnically matched control individuals.
DNA from the affected sisters was subjected to whole-exome sequencing. The genotypes of interest were confirmed, and genotypes of the additional family members and the control subjects were determined by Sanger sequencing.
Results revealed a nonsense homozygous mutation in the SYCE1 gene in both affected sisters. The parents and three brothers were heterozygous for the mutation, and an unaffected sister did not carry the mutation. The mutation was not identified in the DNA samples from the 90 control subjects.
"One of my main topics of interest is puberty," said first author Dr. Liat de Vries, professor of medicine at Tel Aviv University. "The clinical presentation of the 2 sisters, out of 11 children of first-degree cousins, was interesting. In each of the girls, POI was expressed differently. One had reached puberty and was almost fully developed but did not have menses. The second, 16 years old, showed no signs of development whatsoever. By identifying the genetic mutation, we saved the family a lot of heartache by presenting evidence that any chance of inducing fertility in these two girls is slight. As bad as the news is, at least they will not spend years on fertility treatments and will instead invest efforts in acquiring an egg donation, for example. Knowledge is half the battle—and now the entire family knows it should undergo genetic testing for this mutation."
The paper was published in the July 25, 2014, online edition of the Journal of Clinical Endocrinology and Metabolism.
Related Links:
Tel Aviv University
Mammals and many other organisms achieve reduction and recombination through a process called meiosis, which is recognizable by the presence of a distinctive structure—the synaptonemal complex (SCS), —that links the chromosomes together and is essential for meiosis to complete. The protein encoded by the SYCE1 gene (synaptonemal complex central element 1) may have a role in the synaptonemal complex assembly, stabilization, and recombination. Genes involved in essential steps in chromosome synapsis and recombinations during meiosis, such as SYCE1, have been shown to cause POI in animal models but not in humans.
Investigators at Tel Aviv University (Israel) worked with two daughters of consanguineous parents (first cousins) from a 13-member Israeli-Arab family who had been diagnosed with POI. Genotyping was performed in the patients, their parents, four unaffected siblings, and a group of 90 ethnically matched control individuals.
DNA from the affected sisters was subjected to whole-exome sequencing. The genotypes of interest were confirmed, and genotypes of the additional family members and the control subjects were determined by Sanger sequencing.
Results revealed a nonsense homozygous mutation in the SYCE1 gene in both affected sisters. The parents and three brothers were heterozygous for the mutation, and an unaffected sister did not carry the mutation. The mutation was not identified in the DNA samples from the 90 control subjects.
"One of my main topics of interest is puberty," said first author Dr. Liat de Vries, professor of medicine at Tel Aviv University. "The clinical presentation of the 2 sisters, out of 11 children of first-degree cousins, was interesting. In each of the girls, POI was expressed differently. One had reached puberty and was almost fully developed but did not have menses. The second, 16 years old, showed no signs of development whatsoever. By identifying the genetic mutation, we saved the family a lot of heartache by presenting evidence that any chance of inducing fertility in these two girls is slight. As bad as the news is, at least they will not spend years on fertility treatments and will instead invest efforts in acquiring an egg donation, for example. Knowledge is half the battle—and now the entire family knows it should undergo genetic testing for this mutation."
The paper was published in the July 25, 2014, online edition of the Journal of Clinical Endocrinology and Metabolism.
Related Links:
Tel Aviv University
Latest Molecular Diagnostics News
- Game-Changing Blood Test for Stroke Detection Could Bring Life-Saving Care to Patients
- Blood Proteins Could Warn of Cancer Seven Years before Diagnosis
- New DNA Origami Technique to Advance Disease Diagnosis
- Ultrasound-Aided Blood Testing Detects Cancer Biomarkers from Cells
- New Respiratory Syndromic Testing Panel Provides Fast and Accurate Results
- New Synthetic Biomarker Technology Differentiates Between Prior Zika and Dengue Infections
- Novel Biomarkers to Improve Diagnosis of Renal Cell Carcinoma Subtypes
- RNA-Powered Molecular Test to Help Combat Early-Age Onset Colorectal Cancer
- Advanced Blood Test to Spot Alzheimer's Before Progression to Dementia
- Multi-Omic Noninvasive Urine-Based DNA Test to Improve Bladder Cancer Detection
- First of Its Kind NGS Assay for Precise Detection of BCR::ABL1 Fusion Gene to Enable Personalized Leukemia Treatment
- Urine Test to Revolutionize Lyme Disease Testing
- Simple Blood Test Could Enable First Quantitative Assessments for Future Cerebrovascular Disease
- New Genetic Testing Procedure Combined With Ultrasound Detects High Cardiovascular Risk
- Blood Samples Enhance B-Cell Lymphoma Diagnostics and Prognosis
- Blood Test Predicts Knee Osteoarthritis Eight Years Before Signs Appears On X-Rays
Channels
Clinical Chemistry
view channel
3D Printed Point-Of-Care Mass Spectrometer Outperforms State-Of-The-Art Models
Mass spectrometry is a precise technique for identifying the chemical components of a sample and has significant potential for monitoring chronic illness health states, such as measuring hormone levels... Read more.jpg)
POC Biomedical Test Spins Water Droplet Using Sound Waves for Cancer Detection
Exosomes, tiny cellular bioparticles carrying a specific set of proteins, lipids, and genetic materials, play a crucial role in cell communication and hold promise for non-invasive diagnostics.... Read more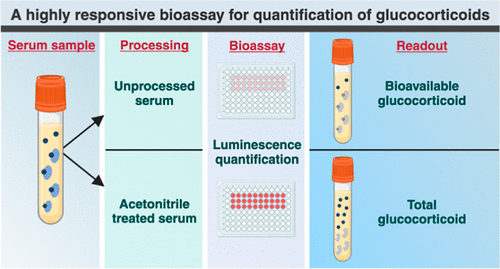
Highly Reliable Cell-Based Assay Enables Accurate Diagnosis of Endocrine Diseases
The conventional methods for measuring free cortisol, the body's stress hormone, from blood or saliva are quite demanding and require sample processing. The most common method, therefore, involves collecting... Read moreHematology
view channel
Next Generation Instrument Screens for Hemoglobin Disorders in Newborns
Hemoglobinopathies, the most widespread inherited conditions globally, affect about 7% of the population as carriers, with 2.7% of newborns being born with these conditions. The spectrum of clinical manifestations... Read more
First 4-in-1 Nucleic Acid Test for Arbovirus Screening to Reduce Risk of Transfusion-Transmitted Infections
Arboviruses represent an emerging global health threat, exacerbated by climate change and increased international travel that is facilitating their spread across new regions. Chikungunya, dengue, West... Read more
POC Finger-Prick Blood Test Determines Risk of Neutropenic Sepsis in Patients Undergoing Chemotherapy
Neutropenia, a decrease in neutrophils (a type of white blood cell crucial for fighting infections), is a frequent side effect of certain cancer treatments. This condition elevates the risk of infections,... Read more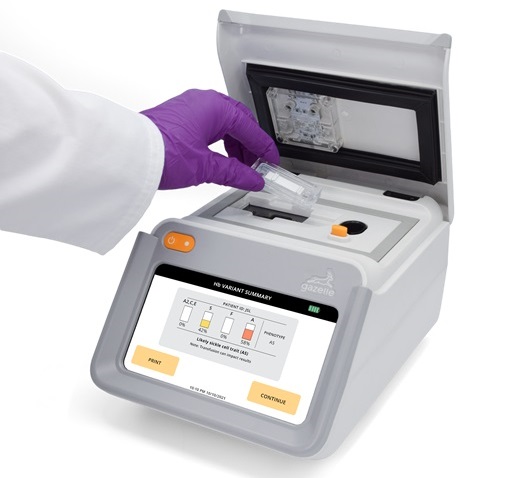
First Affordable and Rapid Test for Beta Thalassemia Demonstrates 99% Diagnostic Accuracy
Hemoglobin disorders rank as some of the most prevalent monogenic diseases globally. Among various hemoglobin disorders, beta thalassemia, a hereditary blood disorder, affects about 1.5% of the world's... Read moreImmunology
view channel.jpg)
AI Predicts Tumor-Killing Cells with High Accuracy
Cellular immunotherapy involves extracting immune cells from a patient's tumor, potentially enhancing their cancer-fighting capabilities through engineering, and then expanding and reintroducing them into the body.... Read more
Diagnostic Blood Test for Cellular Rejection after Organ Transplant Could Replace Surgical Biopsies
Transplanted organs constantly face the risk of being rejected by the recipient's immune system which differentiates self from non-self using T cells and B cells. T cells are commonly associated with acute... Read more
AI Tool Precisely Matches Cancer Drugs to Patients Using Information from Each Tumor Cell
Current strategies for matching cancer patients with specific treatments often depend on bulk sequencing of tumor DNA and RNA, which provides an average profile from all cells within a tumor sample.... Read more
Genetic Testing Combined With Personalized Drug Screening On Tumor Samples to Revolutionize Cancer Treatment
Cancer treatment typically adheres to a standard of care—established, statistically validated regimens that are effective for the majority of patients. However, the disease’s inherent variability means... Read moreMicrobiology
view channel
Integrated Solution Ushers New Era of Automated Tuberculosis Testing
Tuberculosis (TB) is responsible for 1.3 million deaths every year, positioning it as one of the top killers globally due to a single infectious agent. In 2022, around 10.6 million people were diagnosed... Read more
Automated Sepsis Test System Enables Rapid Diagnosis for Patients with Severe Bloodstream Infections
Sepsis affects up to 50 million people globally each year, with bacteraemia, formerly known as blood poisoning, being a major cause. In the United States alone, approximately two million individuals are... Read moreEnhanced Rapid Syndromic Molecular Diagnostic Solution Detects Broad Range of Infectious Diseases
GenMark Diagnostics (Carlsbad, CA, USA), a member of the Roche Group (Basel, Switzerland), has rebranded its ePlex® system as the cobas eplex system. This rebranding under the globally renowned cobas name... Read more
Clinical Decision Support Software a Game-Changer in Antimicrobial Resistance Battle
Antimicrobial resistance (AMR) is a serious global public health concern that claims millions of lives every year. It primarily results from the inappropriate and excessive use of antibiotics, which reduces... Read morePathology
view channel
New AI Tool Classifies Brain Tumors More Quickly and Accurately
Precision in diagnosing and categorizing tumors is essential for delivering effective treatment to patients. Currently, the gold standard for identifying various types of brain tumors involves DNA methylation-based... Read more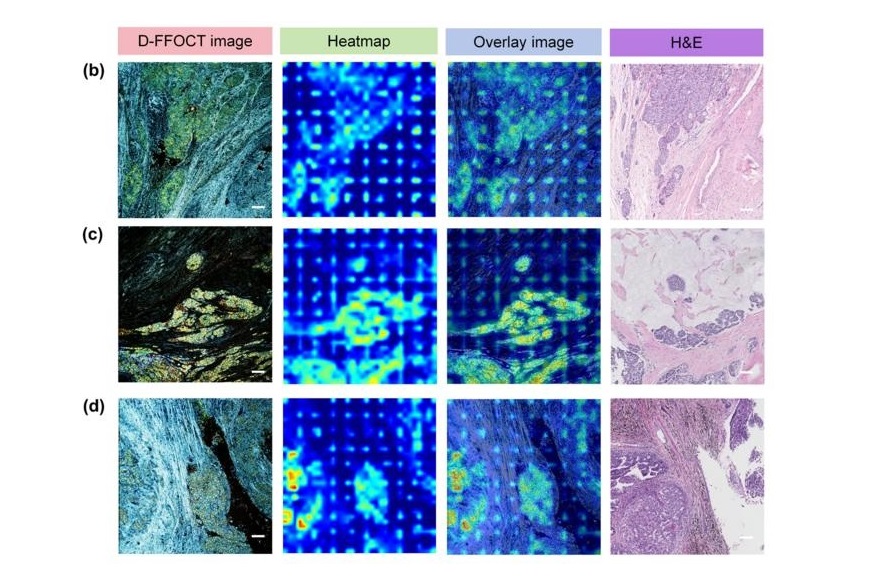
AI Integrated With Optical Imaging Technology Enables Rapid Intraoperative Diagnosis
Rapid and accurate intraoperative diagnosis is essential for tumor surgery as it guides surgical decisions with precision. Traditional intraoperative assessments, such as frozen sections based on H&E... Read more
HPV Self-Collection Solution Improves Access to Cervical Cancer Testing
Annually, over 604,000 women across the world are diagnosed with cervical cancer, and about 342,000 die from this disease, which is preventable and primarily caused by the Human Papillomavirus (HPV).... Read moreHyperspectral Dark-Field Microscopy Enables Rapid and Accurate Identification of Cancerous Tissues
Breast cancer remains a major cause of cancer-related mortality among women. Breast-conserving surgery (BCS), also known as lumpectomy, is the removal of the cancerous lump and a small margin of surrounding tissue.... Read moreTechnology
view channel
New Diagnostic System Achieves PCR Testing Accuracy
While PCR tests are the gold standard of accuracy for virology testing, they come with limitations such as complexity, the need for skilled lab operators, and longer result times. They also require complex... Read more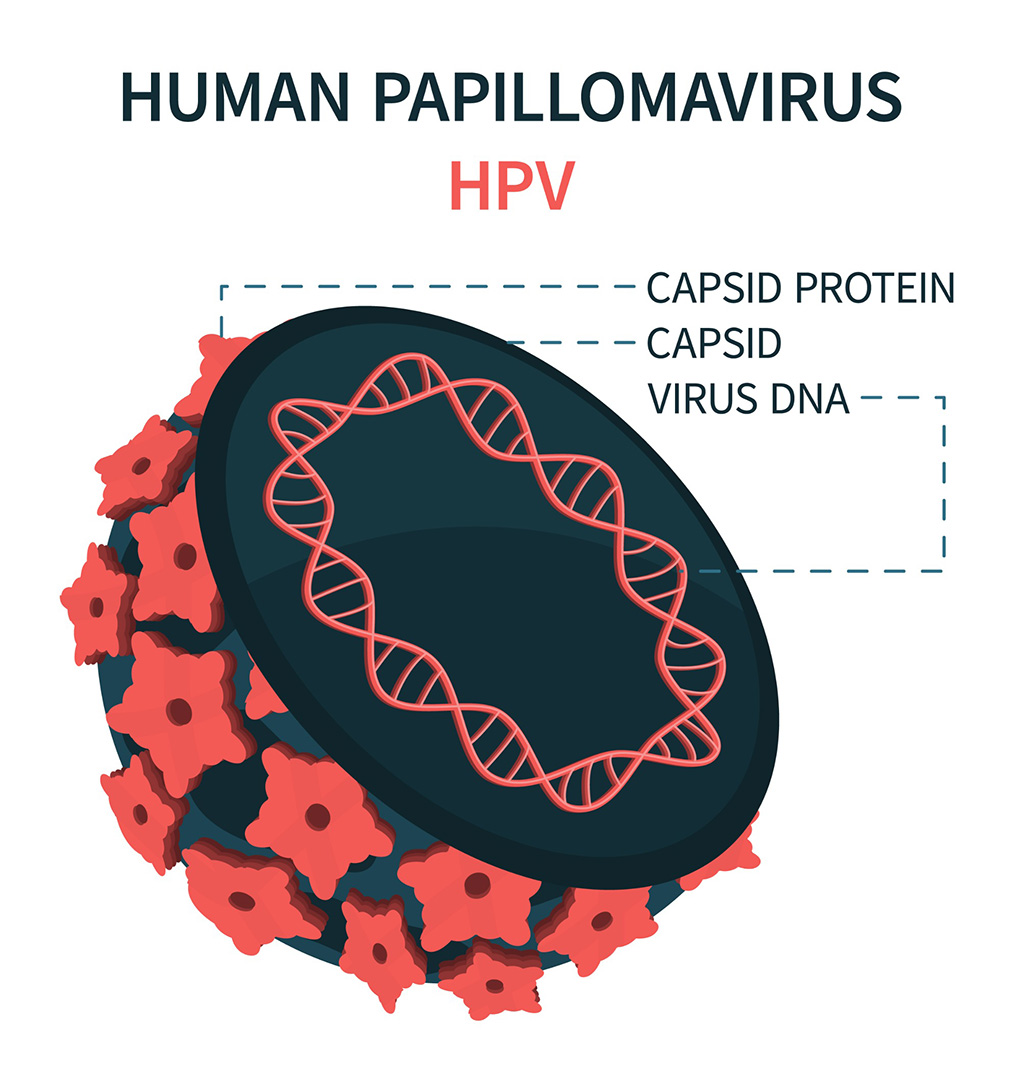
DNA Biosensor Enables Early Diagnosis of Cervical Cancer
Molybdenum disulfide (MoS2), recognized for its potential to form two-dimensional nanosheets like graphene, is a material that's increasingly catching the eye of the scientific community.... Read more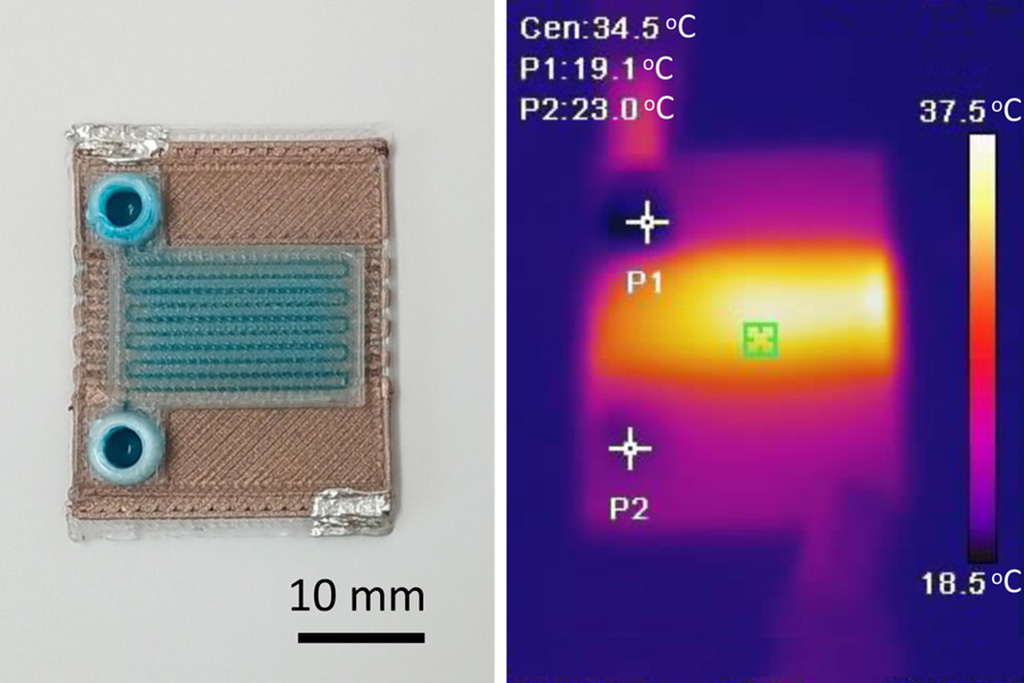
Self-Heating Microfluidic Devices Can Detect Diseases in Tiny Blood or Fluid Samples
Microfluidics, which are miniature devices that control the flow of liquids and facilitate chemical reactions, play a key role in disease detection from small samples of blood or other fluids.... Read more
Breakthrough in Diagnostic Technology Could Make On-The-Spot Testing Widely Accessible
Home testing gained significant importance during the COVID-19 pandemic, yet the availability of rapid tests is limited, and most of them can only drive one liquid across the strip, leading to continued... Read moreIndustry
view channel
Danaher and Johns Hopkins University Collaborate to Improve Neurological Diagnosis
Unlike severe traumatic brain injury (TBI), mild TBI often does not show clear correlations with abnormalities detected through head computed tomography (CT) scans. Consequently, there is a pressing need... Read more
Beckman Coulter and MeMed Expand Host Immune Response Diagnostics Partnership
Beckman Coulter Diagnostics (Brea, CA, USA) and MeMed BV (Haifa, Israel) have expanded their host immune response diagnostics partnership. Beckman Coulter is now an authorized distributor of the MeMed... Read more_1.jpg)












