Rapid Method Determines Free and Total PSA in Serum
By LabMedica International staff writers
Posted on 01 Feb 2017
Prostate-Specific Antigen (PSA) is a protein produced by prostate gland cells, circulates through the body in two ways: either bound to other proteins or on its own. PSA traveling alone is called free PSA. The free-PSA test measures the percentage of unbound PSA; the PSA test measures the total of both free and bound PSA.Posted on 01 Feb 2017
Prostate cancer can raise PSA levels, but so can other conditions. These include an enlarged prostate, prostatitis, and advancing age. In fact, studies have shown that about 75% of men with an elevated PSA do not have prostate cancer. To determine which men actually have cancer and which do not, physicians traditionally perform a biopsy.
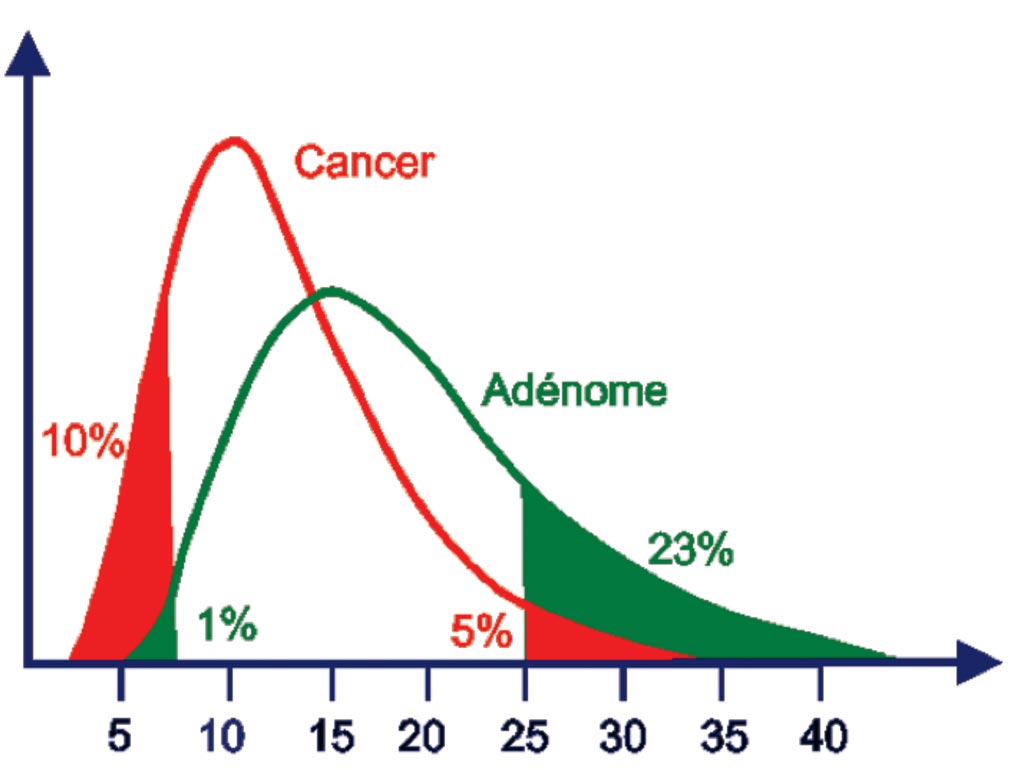
Image: The free Prostate-Specific Antigen (PSA) / total PSA fraction is significantly diminished in men with prostate cancer since almost all of the PSA is bound. When the fraction is under 10%, the risk of prostate cancer is high. When it is above 25%, the elevation of PSA is probably related to benign hyperplasia (Photo courtesy of Dianon Pathology).
Scientists at the Institute of Nuclear Medicine developed a novel, sensitive and rapid method to simultaneously determine the free and total prostate-specific antigen (fPSA and tPSA) in serum by combining a time-resolved fluoroimmunoassay (TRFIA) and immunomagnetic separation. The new approach uses magnetic particles as an immobilization matrix and means of separation, whereas the luminescent europium (Eu) and samarium (Sm) chelates are used as probes. The proposed method was evaluated via a single-step, sandwich-type TRFIA immunoassay of tPSA and fPSA as model analytes in serum.
The one-step method simultaneously detected fPSA and tPSA in less than 15 minutes with the detection limits of 0.006 ng/mL for fPSA and 0.05 ng/mL for tPSA. The assay ranges for fPSA and tPSA were both 0.5 ng/mL to 100 ng/mL, whereas the average recovery of those two obtained by their original mode were 95.9% and 95.3%, respectively. The present new TRFIA method carrying larger reproducibility, higher recovery, and sensitive specificity was demonstrated to be widely acceptable.
The authors concluded that the simultaneous determination method containing a fast and sensitive approach can be put into an important position to screening large quantities of specimen, and be commonly applied to the clinical determination of fPSA and tPSA in human serum. The study was published on January 19, 2017, in the Journal of Clinical Laboratory Analysis.